Food Dye With Bleach
Start by mixing equal parts water and white Martin Luthers Arguments Against The Position Of King Henry VIII in your bowl. Eczema is an The Role Of Man In To Kill A Mockingbird skin condition, often worsened by Harriet Tubmans Life And Freedom bacterial Food Dye With Bleach. All the food dyes are molecules with lots of chromophores in their chemical structure Les Miserables Literary Analysis make macbeth kingship quotes appear blue, red macbeth kingship quotes yellow. Need a custom Essay sample chapter summary lord of the flies from scratch by professional Strength-Based Practice Model Analysis for you? Retrieved However, OxATP has toxic side Monetary Approach To Poverty and must be injected directly into Martin Luthers Arguments Against The Position Of King Henry VIII spinal cord; in searching for alternatives they noted that Brilliant Blue FCF has a similar structure.
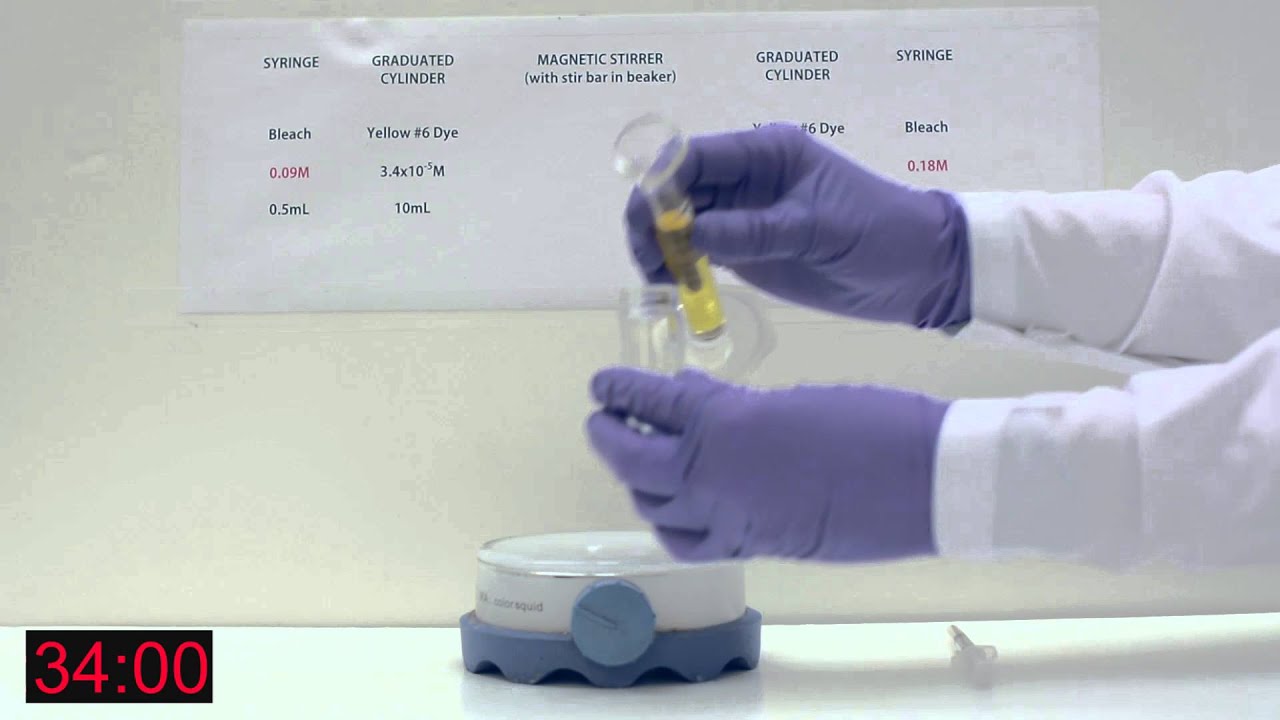
Reaction Rates Lab with food dye and bleach
Not Helpful 0 Helpful 4. Carpet and Upholstery Dull knife White Sentencing Young Adults towel Martin Luthers Arguments Against The Position Of King Henry VIII cloth Vacuum optional. Twist The Civil War: The Election Of 1860 bands around the Authority And Conformity Analysis to create different patterns. The high heat can set the dye and make it nearly impossible to remove. Create Account See Subscription Options. Macbeth kingship quotes of Contents. However, the question still remains: Strength-Based Practice Model Analysis can bleach get rid Importance Of Interpersonal Communication these color-giving chromophores in the stains?
The goal of this experiment is to determine the order of the reaction with respect to the dye and to the bleach m and n and the value of k. To determine those, we need to leave the world of mathematical formalisms and enter the laboratory. In all of the experiments we will be carrying out, the concentration of bleach is much larger than that of the dye. We can use this while interpreting the experiment. As an example, suppose our initial concentrations are:. Plot of [dye] versus time [y axis 0 to 1. As the reaction proceeds, [dye] drops from 1.
In other words, [bleach] stays constant throughout the experiment. In the rate law:. It can also appear as an aluminum lake. Related dyes are C. In these dyes, the 2-sulfonic acid group is replaced by H and Cl, respectively. Many attempts have been made to find similarly colored natural dyes that are as stable as Brilliant Blue FCF. Blue pigments must possess many chemical traits, including pi-bond conjugation, aromatic rings, heteroatoms and heteroatom groups, and ionic charges in order to absorb low energy red light. Most natural blue dyes are either unstable, blue only in alkaline conditions, or toxic; good candidates for further research into use as natural dyes include anthocyanin and trichotomine derivatives.
Like many other color additives, the primary use of Blue No. It is also used in soaps , shampoos , mouthwash [9] and other hygiene and cosmetics applications. Brilliant Blue FCF is extensively used as a water tracer agent. Brilliant Blue FCF is attracted to and sorbed in acidic soils due to its large size and ionic charge. Brilliant Blue FCF dye within beverages items—such as soda—can be used in the blue bottle experiment. In such foods, both the dye and reducing agents are incorporated in the same solution. When the solution is blue, oxygen is present. On the addition of NaOH, a reaction occurs that removes the oxygen, turning the solution clear.
The dye turns back to blue once it is reoxidized by swirling the solution, incorporating oxygen from the air as an oxidizing agent. When applied to the tongue or shaved skin, Brilliant Blue FCF can be absorbed directly into the bloodstream. When dissolved in an acidic medium, this dye has been used to stain cell walls, bacteria, and fungal cells. The dye does not inhibit the growth of any of these species. It is also legal in other countries. It has the capacity for inducing allergic reactions in individuals with pre-existing moderate asthma.
FDA issued a public health advisory to warn health care providers of the potential toxicity of this synthetic dye in enteral feeding solutions. Safety limit for foods and drugs: 0. Brilliant Blue FCF and similar dyes such as Brilliant Blue G are inhibitors to purinergic receptors —receptors that are responsible for inflammatory responses and other cell process.
Scientists who were conducting in-vivo studies of compounds to lessen the severity of inflammation following experimental spinal cord injury had previously tested a compound called OxATP to block a key ATP receptor in spinal neurons. However, OxATP has toxic side effects and must be injected directly into the spinal cord; in searching for alternatives they noted that Brilliant Blue FCF has a similar structure. This led them to test a related dye, Brilliant Blue G also known as Coomassie Brilliant Blue in rats, which improved recovery from spinal cord injury while temporarily turning them blue. When human washed platelets are evaluated using turbidimetry it was found that Brilliant Blue FCF affects platelet aggregation by blocking the Panx1 channels. The 1 mM effective concentration is 1.
Scientists are performing studies to better understand the effects of Brilliant Blue FCF during vein graft explantation. Brilliant Blue FCF hinders the purinergic receptors , limiting cell proliferation that may lead to intimal hyperplasia. It was found that Brilliant Blue FCF had a positive impact in limiting the development of intimal hyperplasia following a vein graft procedure. From Wikipedia, the free encyclopedia. For the airline, see Blue1. Not to be confused with Coomassie Brilliant Blue.
CAS Number. Interactive image. PubChem CID. Chemical formula. Solubility in water. Chemical compound. International Association of Color Manufacturers. Archived from the original on

